|
There does not seem to be any clear overall trend or pattern between melting points and atomic numbers for the period four transition metal elements. Vanadium is less dense than copper and has a smaller atomic radius.Īlthough the trends in the physical properties of transition metals in period 4 are not always obvious or fully consistent across all of the different elements, subtle differences can be seen.įor example, as we move from scandium to copper, the density of the different metals increases however, at the same time, the atomic radius decreases in size, due in part to increasing nuclear charge.Ĭombining these two trends we can identify answer B as the correct answer as vanadium is less dense than copper that has a larger atomic radius.Vanadium is denser than copper and has a larger atomic radius.Vanadium is less dense than copper but has a larger atomic radius.Vanadium is denser than copper but has a smaller atomic radius.Which of the following statements that compare two d-block elements is correct? The following figure uses a combination of condensed notations and single-sided arrows to show how the electron configuration changes across the period four metal elements.Įxample 2: Comparing the Densities of Vanadium and Copper The relationship between atomic radii values and atomic numbers is quite complex and it can only be explained if we consider the electronic configurations of these elements. Atomic radius values do not systematically increase or systematically decrease as we move across the row of period four transition metals. We can also consider how atomic radii change as we move from scandium through to copper. Therefore, the correct answer is answer B. 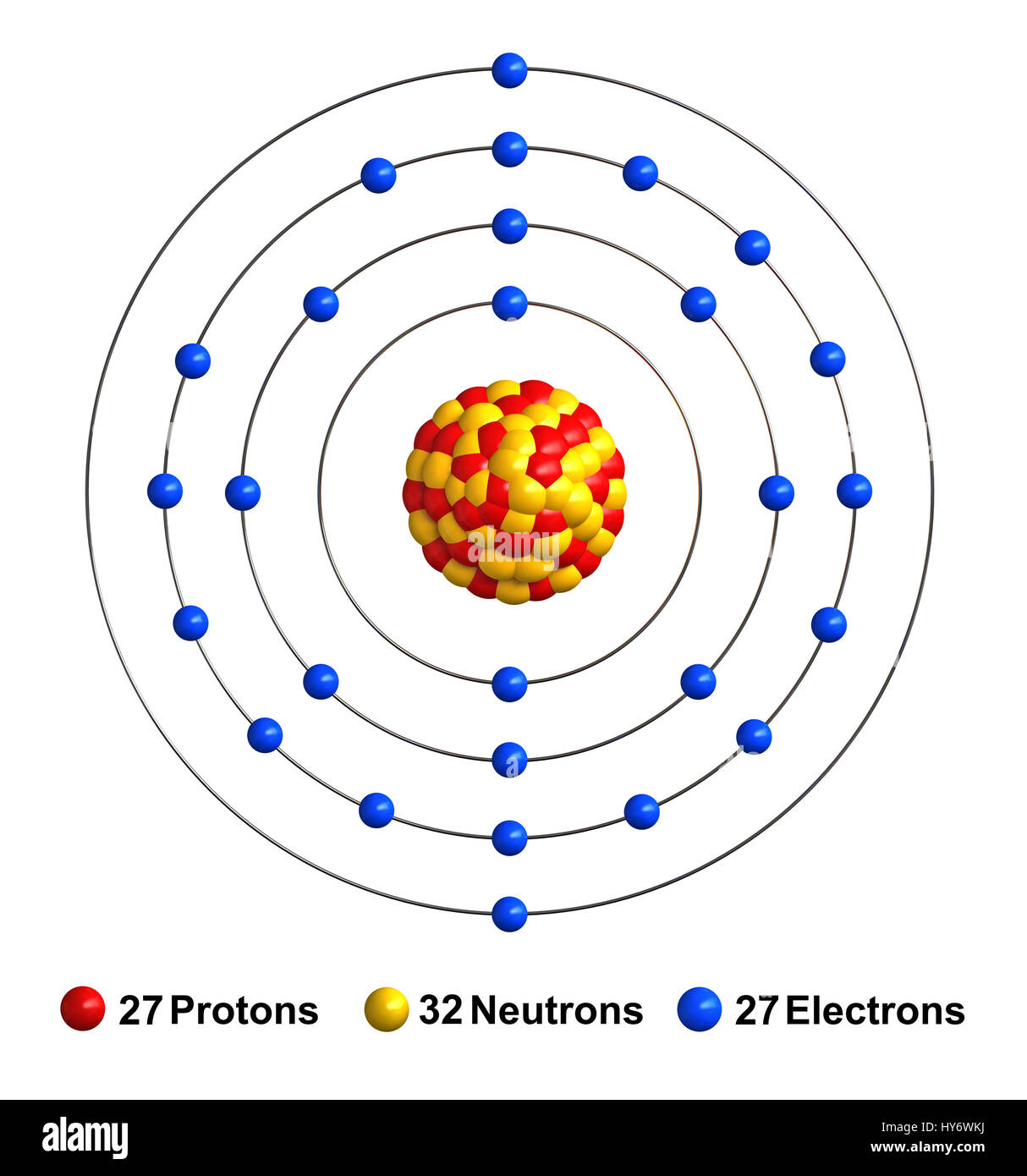 
This trend is seen in the period four transition metals however, nickel with a relative atomic mass of 58.7 is an exception as the preceding element cobalt has an atomic mass of 58.9. 
As the number of protons in the nucleus of the atom increases, so does the number of neutrons creating a general increase in the atomic mass as we move from the left-hand side of the periodic table to the right-hand side. As such, as we proceed across period 4, the atomic number increases, meaning an increase in the number of protons in the nucleus of the different atoms. The elements on the periodic table are ordered in terms of increasing atomic number. Which transition metal of the fourth period is an exception to this trend? The trend in increasing atomic mass is observed across the periods of the periodic table from left to right. Example 1: Selecting Exceptions to Atomic Mass Trends in the Properties of Transition Metals in the Fourth Period
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
